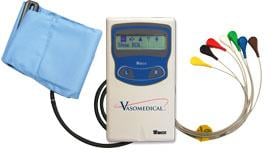
Combined Holter Records Both ECG, Blood Pressure
April 13, 2010 – The U.S. Food and Drug Administration (FDA) recently cleared a combined electrocardiogram (ECG) Holter and ambulatory blood pressure monitoring recorder and software analysis system. The Vasomedical-Biox Model 2301 system is among the first combined systems to simultaneously and continuously record and store ECG and blood pressure data for 24 hours. The ability to view these parameters simultaneously, side by side gives the physician more useful information to evaluate the patient's cardiovascular status. In addition to being a compact device that replaces two separate recorders, it also provides interactive blood pressure recording at times of certain cardiac abnormalities. This can enhance the diagnostic value of the recorded data, while increasing patient comfort and ease of use. For more information: www.vasomedical.com
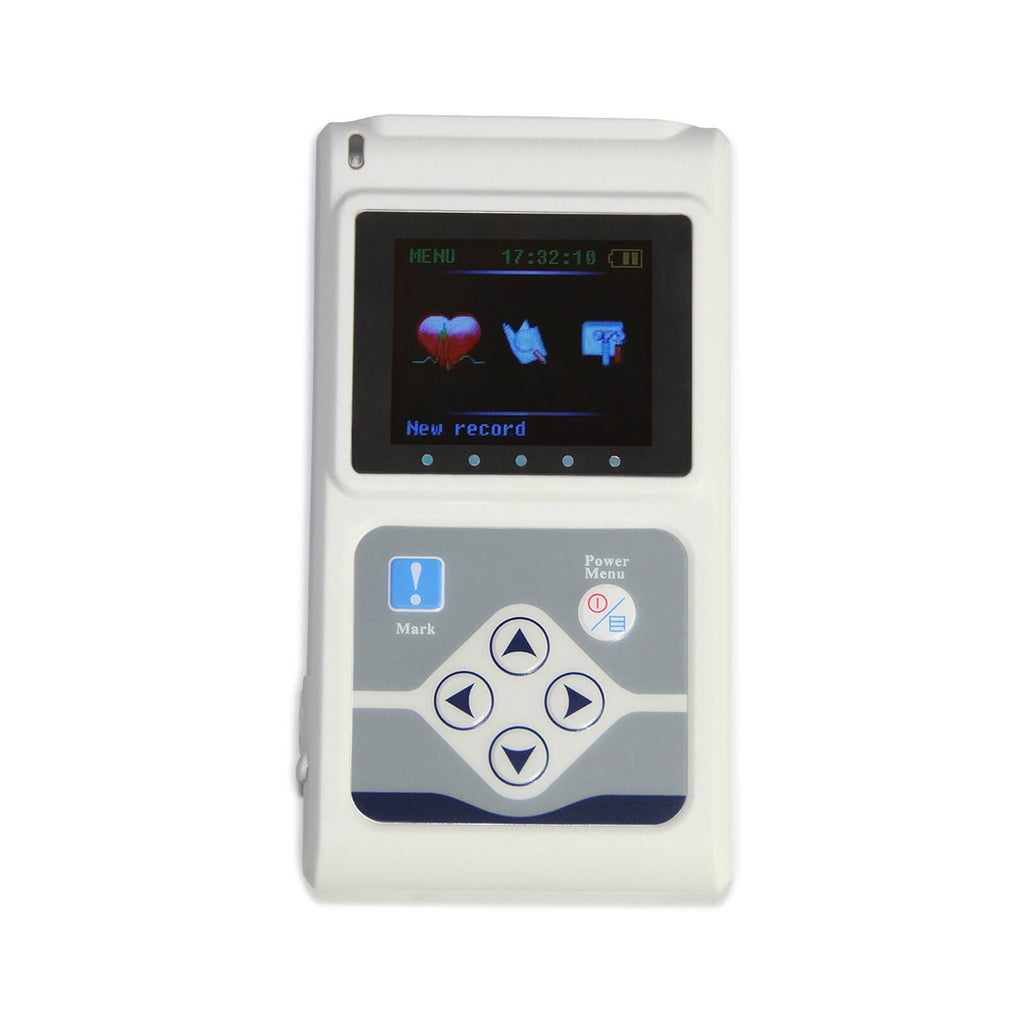
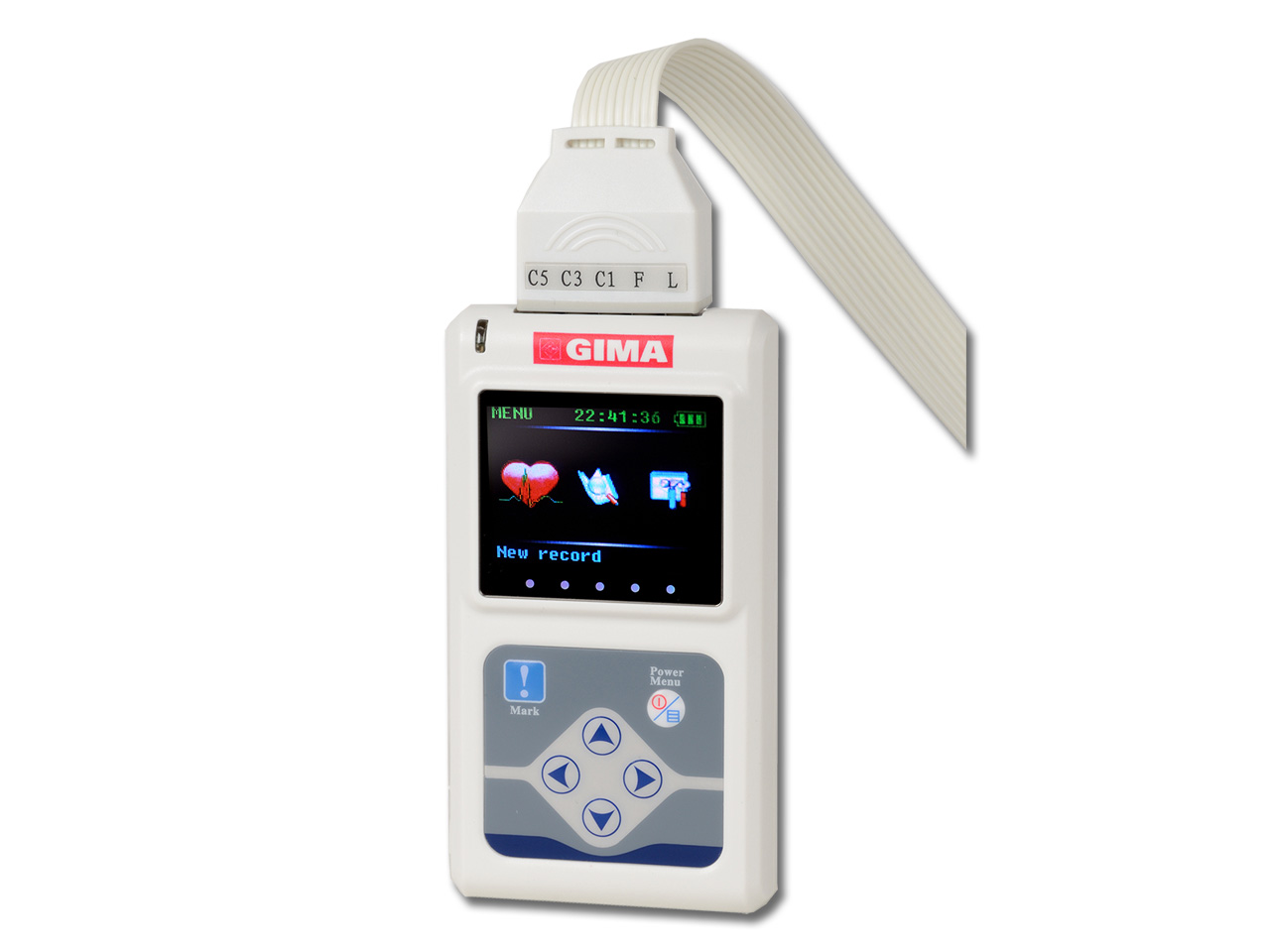
EC-12H 12-Channel Holter ECG System - Biomed Services

Holter Monitoring - an overview

Dynamic 24hours 12 Lead ECG/EKG Holter Monitor Alalyzer Software CONTEC TLC5000 - International Society of Hypertension

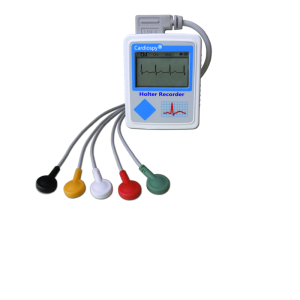
Holter monitor1
:max_bytes(150000):strip_icc()/4172395_color-5bd8c6b346e0fb002d0b0e5e.png)
Holter Monitor Test: Uses, Risks, Procedure, Results
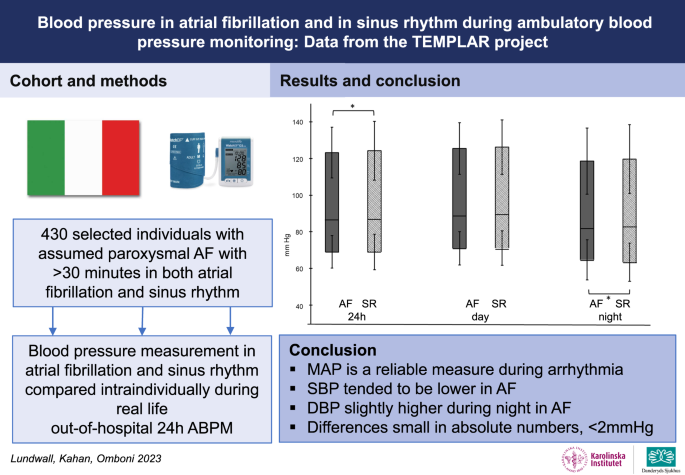
Blood pressure in atrial fibrillation and in sinus rhythm during ambulatory blood pressure monitoring: data from the TEMPLAR project
CHECKME PRO VITAL SIGNS MONITOR WITH ECG HOLTER with Bluetooth

Holter ECG - custo med GmbH - PDF Catalogs

Combined ECG Holter & Ambulatory Blood Pressure Monitor - Vasomedical - PDF Catalogs
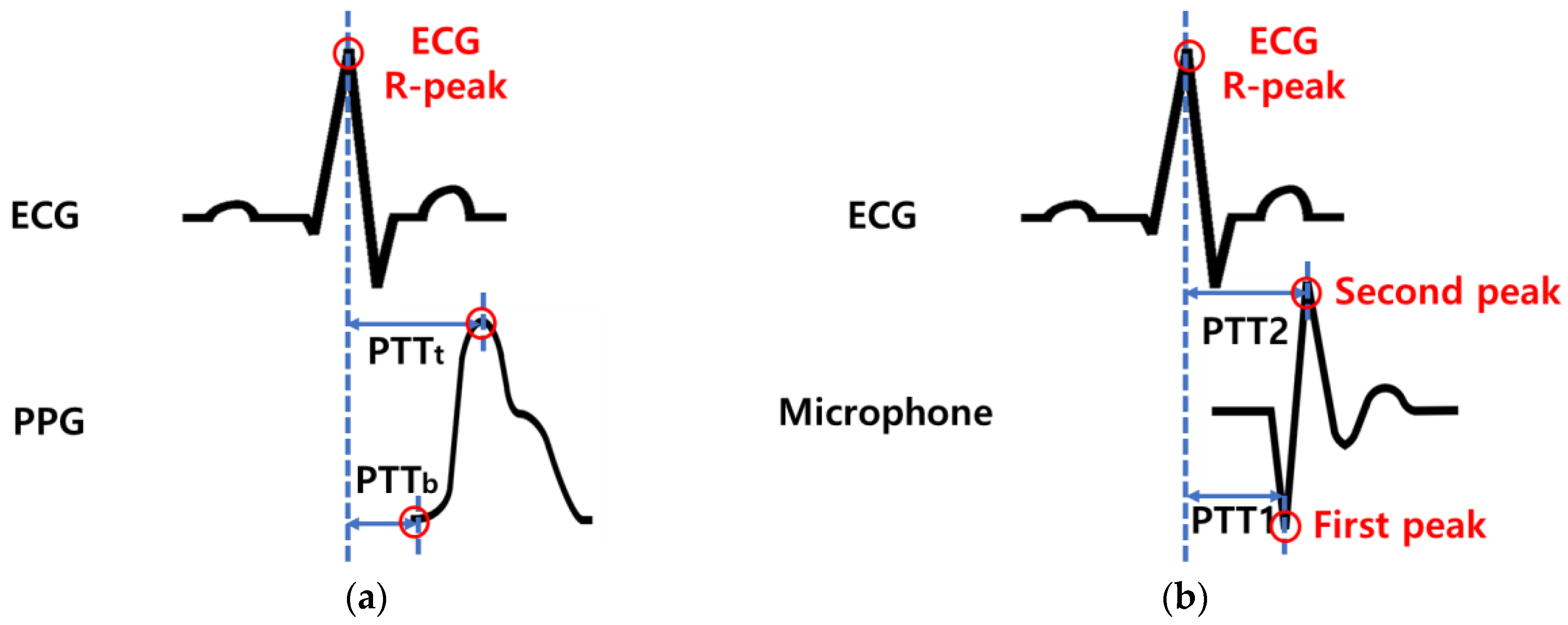
Sensors, Free Full-Text