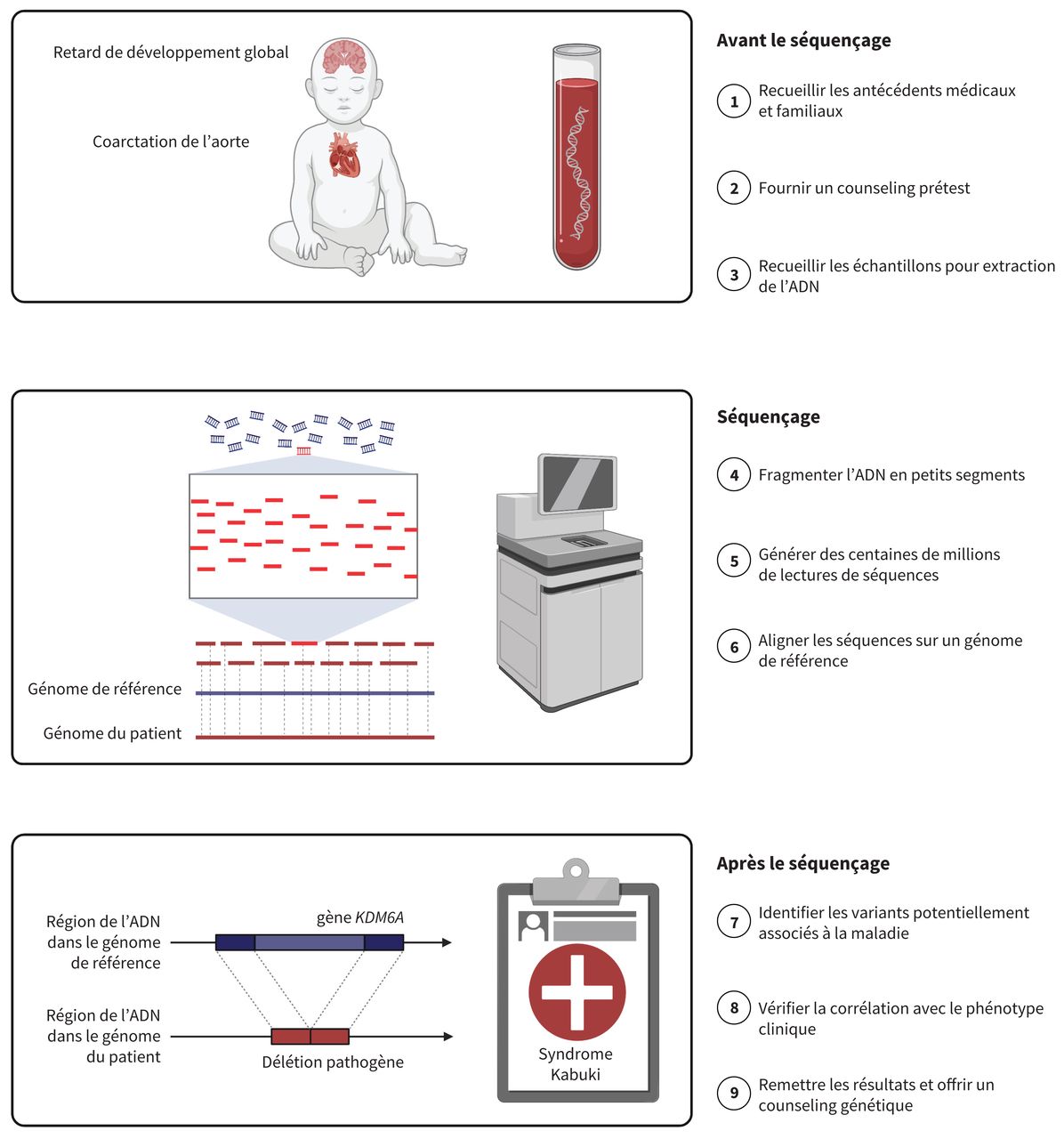
Fabrication de produits de diagnostic in vitro
4.6
(300)
Écrire un avis
Plus
€ 25.50
En Stock
Description

BIOTECanada Insights Spring 2023 by BIOTECanada - Issuu

ProCellics™ Raman Analyzer with Bio4C® PAT Raman Software

Pompes de diagnostic (IVD) In Vitro

Réactifs pour construire votre propre analyse immunologique par

IVD Manufacturing


Montréal InVivo
ISO 18113-2:2022 - This document specifies requirements for information supplied by the manufacturer of in vitro diagnostic (IVD) reagents,

ISO 18113-2:2022 - In vitro diagnostic medical devices — Information supplied by the manufacturer (labelling) — Part 2: In vitro diagnostic reagents f

À propos de nous

In Vitro Diagnostic Regulation (IVDR)
Proposer des recherches
Tu pourrais aussi aimer