![Difference between Strong and Weak Base - with Examples [in Table]](https://d1avenlh0i1xmr.cloudfront.net/0e323ff3-079c-4d5e-b21f-0f4c25082521/differences-between-strong-and-weak-bases-01.jpg)
Difference between Strong and Weak Base - with Examples [in Table]
Strong BaseWeak BaseThey get completely ionized (split up into ions) in water and produce large amounts of hydroxide ions.These only get partially ionized (split up into ions) in water and produce less amount of hydroxide ions.pH value is close to 14 but smaller than it.pH value is closer to 7 but
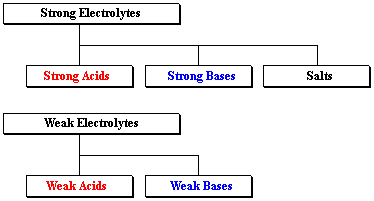
Classifying Electrolytes
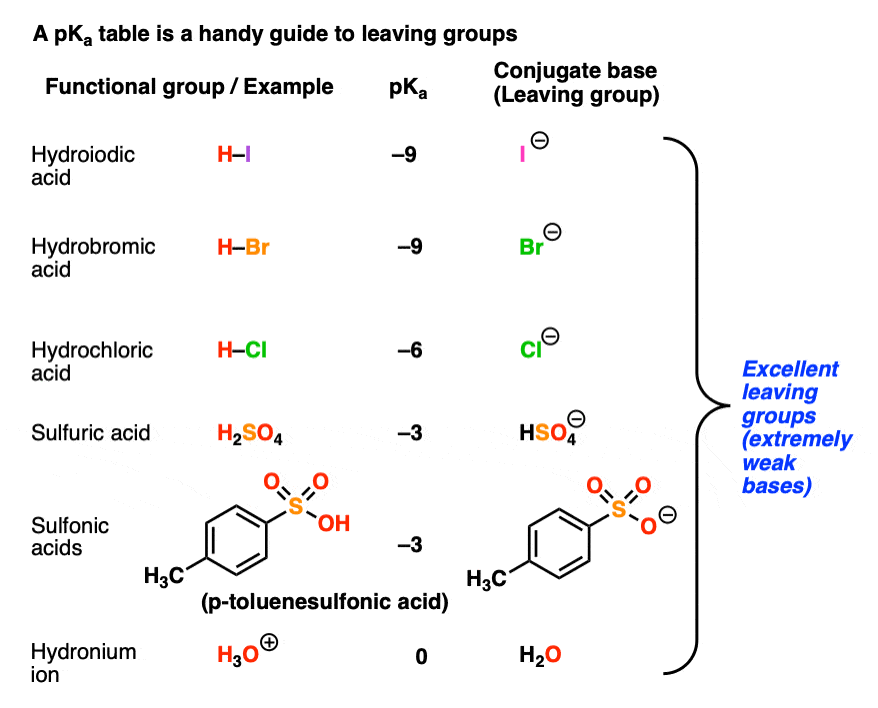
What makes a good leaving group? Master Organic Chemistry
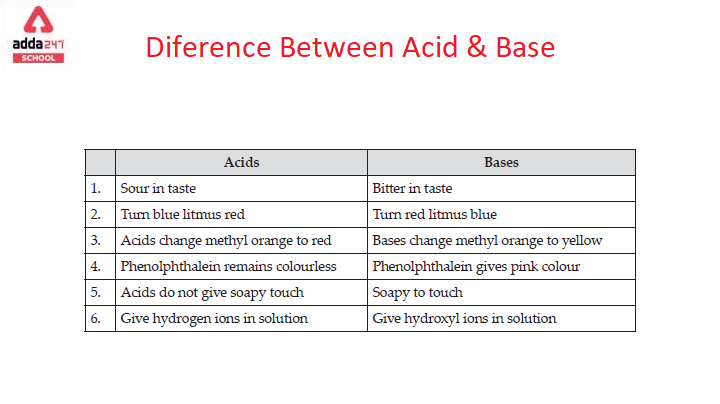
Difference Between Acid and Base for Class 7

2.5: Weak Acids and Weak Bases - Chemistry LibreTexts

Here is an example of a titration curve, produced when a strong base is added to a strong acid. This curve shows how pH varies as 0.100 M NaOH is added to 50.0 mL of 0.100 M HCl.
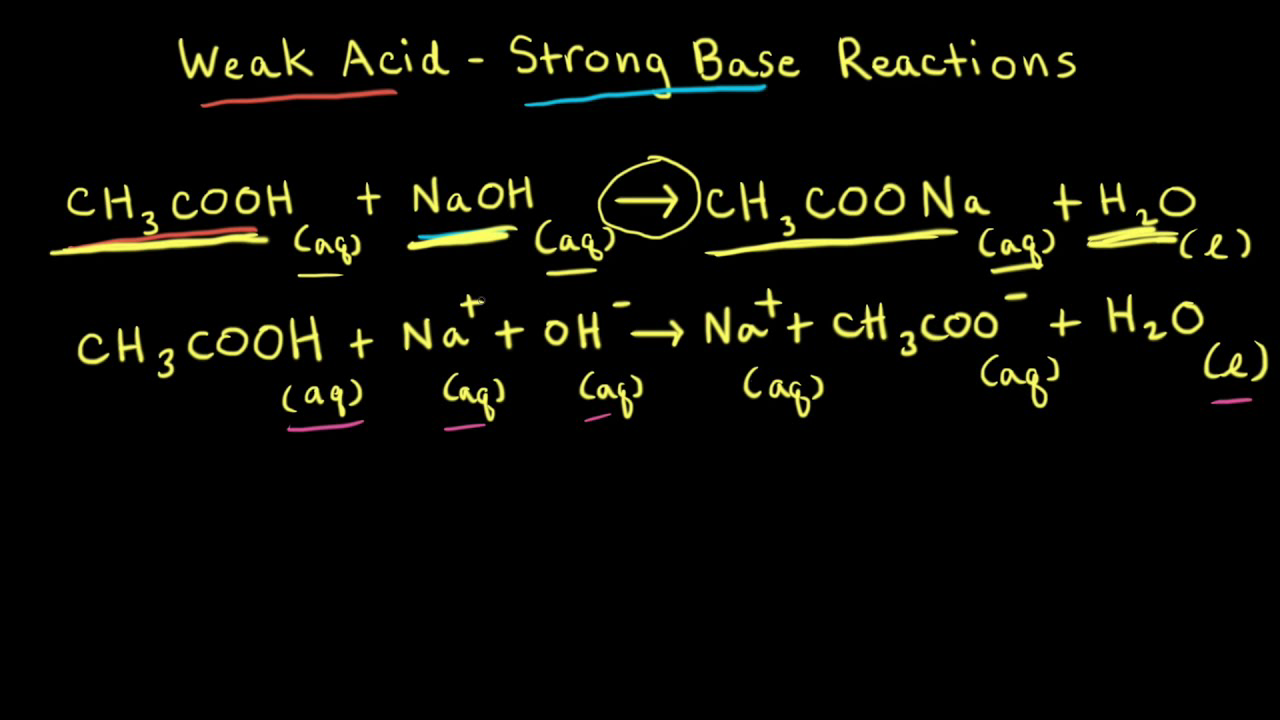
Weak acid–strong base reactions (video)

Strong Base, Overview, Examples & Characteristics - Video & Lesson Transcript

Strong weak acids and bases
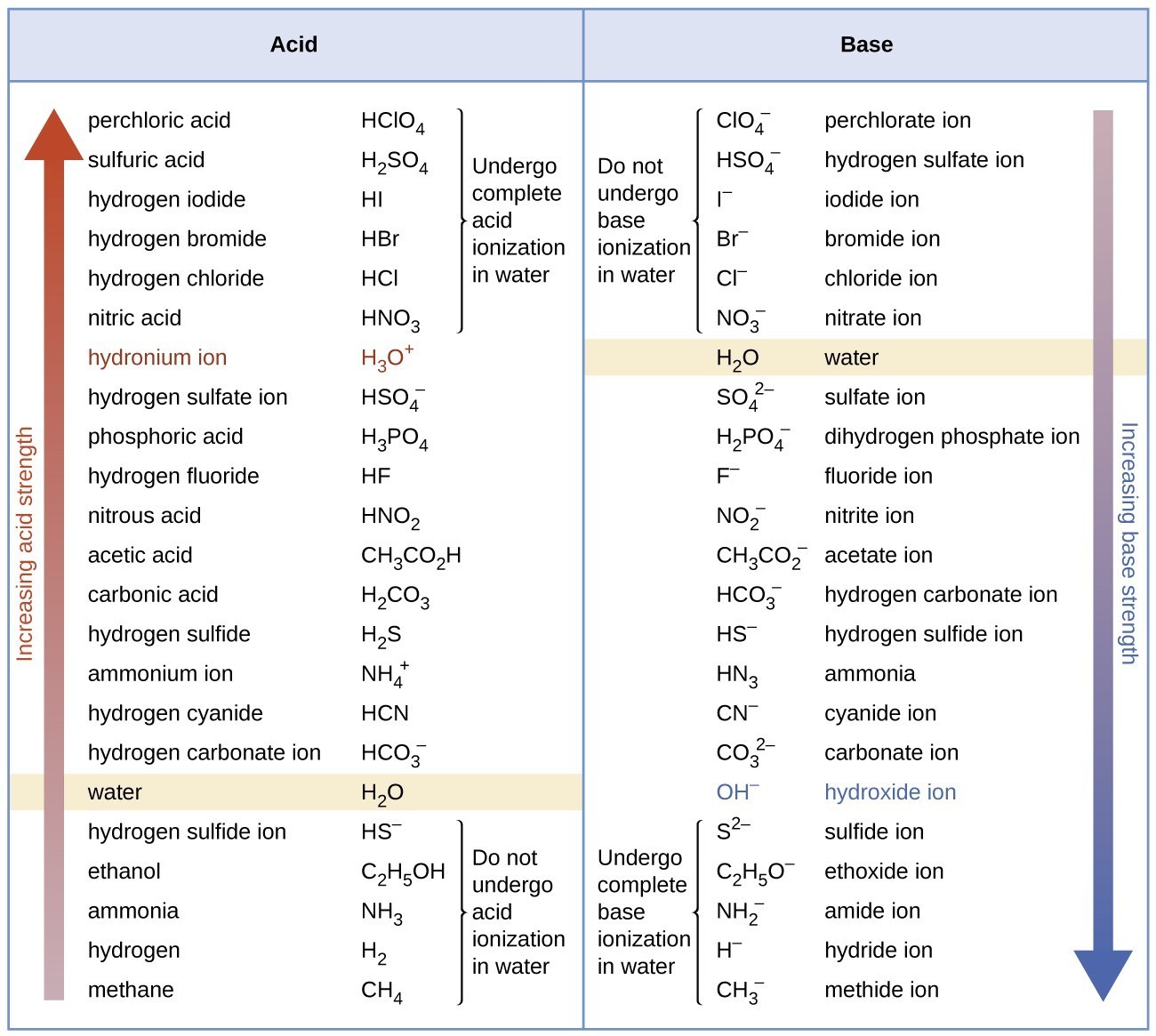
Relative Strengths of Acids and Bases

How To Memorize The Strong Acids and Strong Bases
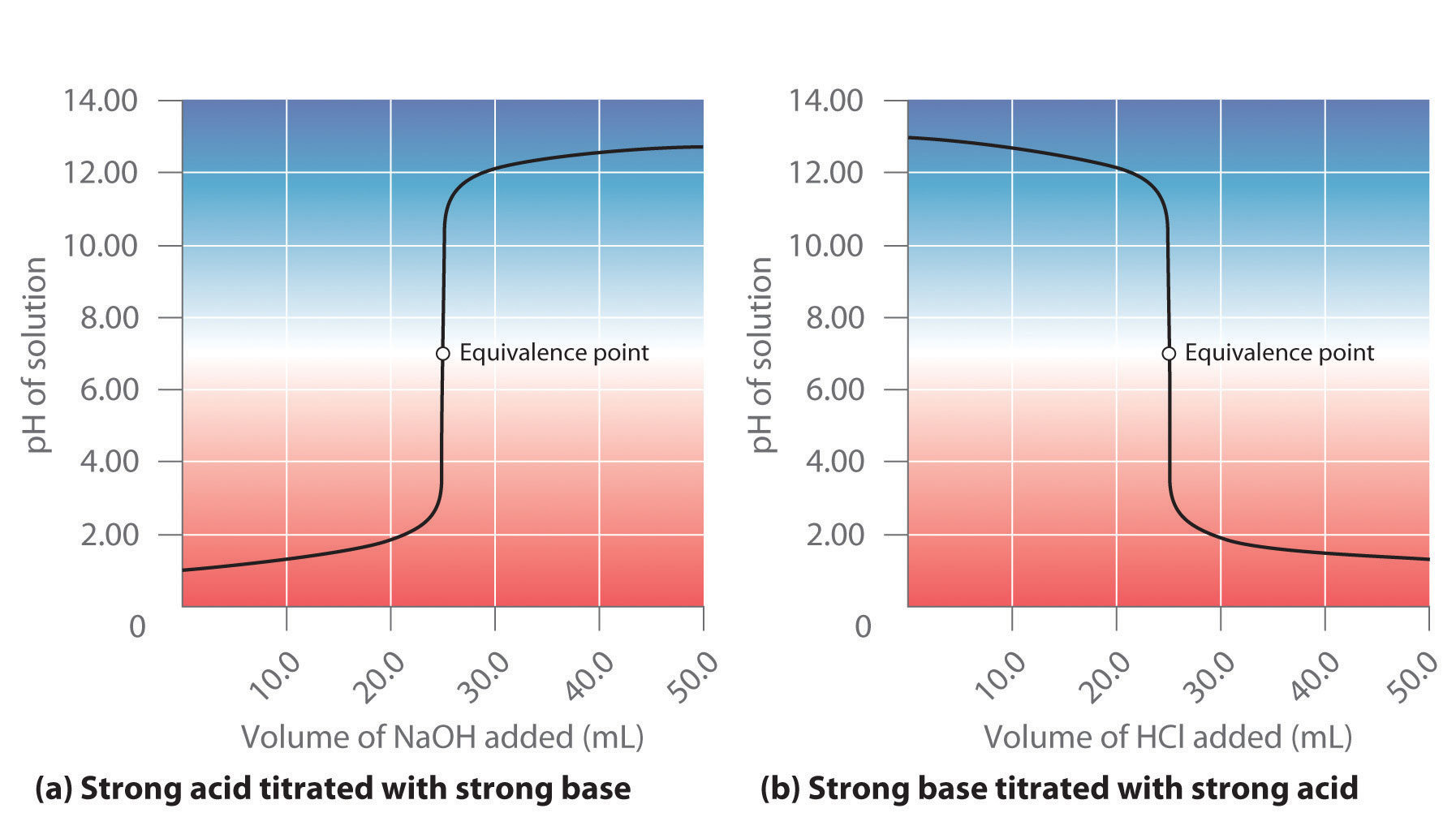
Acid–Base Titrations

Acid Base Titration - Titration Curves, Equivalence Point & Indicators of Acid Base Titration
.png)
What are Bases? - Definition, Examples, Types, Properties and Uses - GeeksforGeeks

Difference between Strong and Weak Base - with Examples [in Table]